For rare disease patients, small patient numbers and the lack of effective treatments are shared challenges for both caregivers and physicians. Over 16 years, the Department of Medical Genetics at National Taiwan University Hospital (NTUH) invested in drug development and clinical trials, achieving the world’s first gene therapy for AADC (aromatic L-amino acid decarboxylase) deficiency.
This therapy has recently received marketing authorization from the European Medicines Agency (EMA), offering a glimmer of hope for gene therapy in rare diseases.
Typically, children develop head control by three months and can sit by six months. However, in Taiwan, 1 in 30,000–50,000 children are born with the congenital metabolic disorder AADC deficiency. Globally, one in three reported cases originates from Taiwan, with most patients concentrated in Taiwan and Southeast Asia.
Children with AADC deficiency lack the enzyme needed to synthesize dopamine, and also have insufficient adrenaline and noradrenaline. Clinically, they exhibit hypotonia, inability to move while lying down, emotional instability, oculogyric crises (upward eye deviation), and autonomic dysfunction. Symptoms can last 6–8 hours or recur every 2–3 days, making feeding and growth difficult. Without treatment, survival beyond age five was rare.
AADC Gene Therapy Delivered via Skull Injection Now Treats Children Under Two
At a 2007 AADC patient gathering organized by the Rare Disease Foundation, a family member with a medical background discovered that Parkinson’s disease patients also show AADC activity loss, causing dopamine deficiency.
Collaborating with Wuh-Liang Hwu, NTU Medical School professor and Distinguished Researcher at China Medical University Hospital’s Precision Medicine Center, the NTUH Medical Genetics Department began investigating the disease mechanism and consulted with Japanese and American experts on therapeutic feasibility.
The Rare Disease Foundation later launched a “Rare Disease Clinical Trial Support Program” to aid local trials. In 2010, NTUH, with Ministry of Health and Welfare approval, completed the world’s first compassionate-use gene therapy for a child with AADC deficiency.
According to Rai-Hseng Hsu, attending physician at NTUH Medical Genetics, the treatment uses an adeno-associated virus (AAV) vector carrying the AADC gene. Early procedures involved placing steel pins on the skull for stereotactic targeting. Nowadays, stereotactic guidance precisely locates injection sites in young children’s skulls, and a robotic arm injects the therapy into the putamen, directly affected by dopamine production deficiency. The therapy can now be applied to children under two years old.
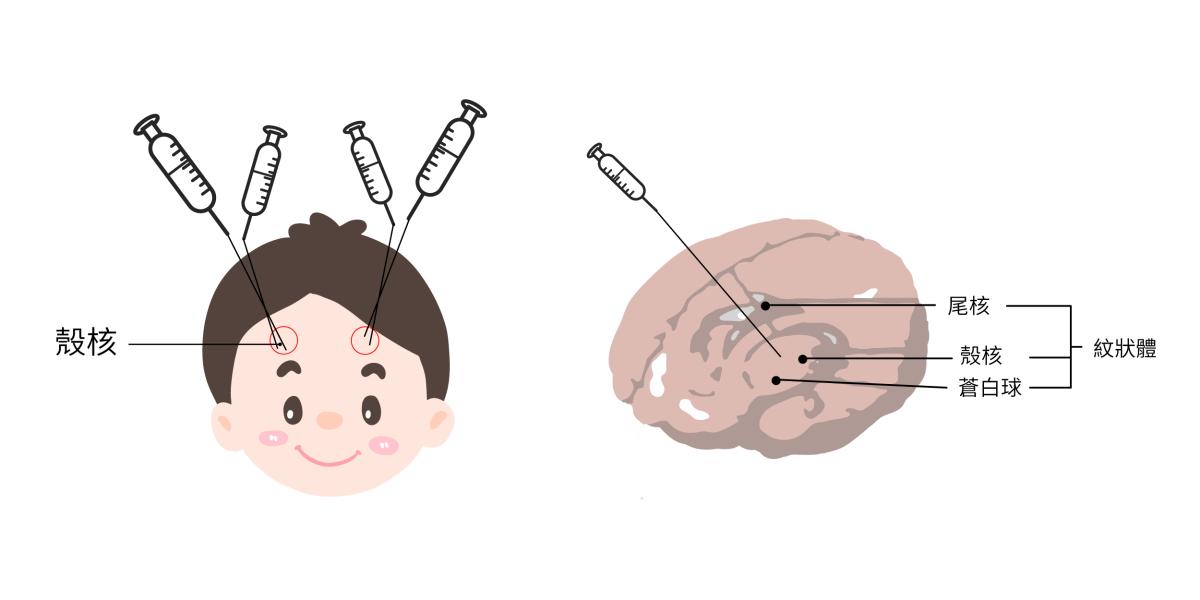
40% of Children Can Walk After Treatment
Yin-Hsiu Chien, Director of NTUH Medical Genetics Department, notes that younger patients respond better. Children are typically diagnosed around age three, though symptoms may appear within the first 2–3 months of life.
NTUH compared outcomes between children treated at 18 months versus eight years old. The younger child could walk, attend school, and speak simple words. Children with milder symptoms and better nutrition also showed greater improvement after early treatment.
After gene therapy, few children develop speech, but approximately 40 percent who previously could not move are able to walk, either independently or with assistance, representing a significant improvement over the previously untreatable condition.
Gene therapy has also spurred the development of the 3-OMD blood spot screening tool. NTUH initiated a pioneering newborn screening program for AADC deficiency, covering 99.5 percent of newborns in Taiwan. The 3-OMD metabolite, a dopamine pathway marker, is elevated in AADC patients, prompting follow-up genetic testing.
Today, NTUH neurologists and neonatologists can differentiate AADC deficiency from cerebral palsy. If confirmed genetically, physicians administer therapy and guide families on potential issues, such as feeding difficulties or low weight, involving physical therapists or nutritionists to support optimal development.
NTUH Expands Technology Transfer to the US, UK, and Europe
Following NTUH gene therapy, five-year survival for treated AADC patients reached 93.5 percent, attracting US biotech company Agilis Biotherapeutics for a collaborative technology transfer.
In 2022, Upstaza™ (generic: Eladocagene exuparvovec) received EMA marketing authorization, becoming the only brain-targeted gene therapy globally approved for AADC deficiency.
NTUH Medical Genetics Department’s achievements are globally recognized. These include earliest treatment, highest patient numbers, youngest patients treated, and safest injection sites. Their work is cited in the pediatric “bible,” Nelson Textbook of Pediatrics (21st Edition).
International competitors are emerging, such as UCSF in the US, which enrolled seven patients in Phase I trials. NTUH continues to support Southeast Asian newborn screening, publishes treatment guidelines, and advocates for insurance coverage. Together, these efforts have maintained the hospital's international impact and transforming the lives of children with rare diseases.
Editorial Note: This article features the Gold Award recipient for Specialty Medical Services, the 25th National Biotechnology and Medicine Care Quality Award. All titles and positions mentioned reflect the interviewees’ roles at the time of the interviews.
